Why fathers don't pass on mitochondria to offspring
Offering insights into a long-standing and mysterious bias in biology, a new study reveals how and why mitochondria are only passed on through a mother's egg -- and not the father's sperm. What's more, experiments from the study show that when paternal mitochondria persist for longer than they should during development, the embryo is at greater risk of lethality.
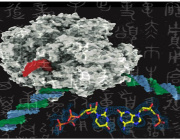
Harbored inside the cells of nearly all multicellular animals, plants and fungi are mitochondria, organelles that play an important role in generating the energy that cells need to survive. Shortly after a sperm penetrates an egg during fertilization, the sperm's mitochondria are degraded while the egg's mitochondria persist. To gain more insights into this highly specific degradation pattern, Qinghua Zhou et al. used electron microscopy and tomography to study sperm mitochondria (or paternal mitochondria) in Caenorhabditis elegans, a type of roundworm, during early stages of development.
Intriguingly, the paternal mitochondria were found to partially self-destruct before the mitochondria were surrounded by autophagosomes, which target components within a cell and facilitate their degradation. This suggests that another mechanism, something within the paternal mitochondrion itself, initiates the degradation process. RNA analysis of paternal mitochondria during early stages of embryonic development hinted that it is the cps-6 gene that facilitates this process, which the team confirmed by studying sperm lacking cps-6; without it, paternal mitochondria remained significantly later into the development stage.
Further investigation suggests that the enzyme that cps-6 encodes first breaks down the interior membrane of the paternal mitochondria before moving to the space within the inner membrane to breakdown mitochondrial DNA. When the researchers engineered paternal mitochondria to breakdown during later stages of development, this increased the chances that the embryo would not survive, suggesting that the transmission of paternal mitochondria is an evolutionary disadvantage.
Collectively, results from this study suggest that cps-6 plays a key role in initiating the self-destruction of paternal sperm, which likely benefits the embryo.
News source: American Association for the Advancement of Science. "Why fathers don't pass on mitochondria to offspring." ScienceDaily. ScienceDaily, 23 June 2016
Journal Reference:
- Qinghua Zhou et al. Mitochondrial endonuclease G mediates breakdown of paternal mitochondria upon fertilization. Science, June 2016